Information for patients
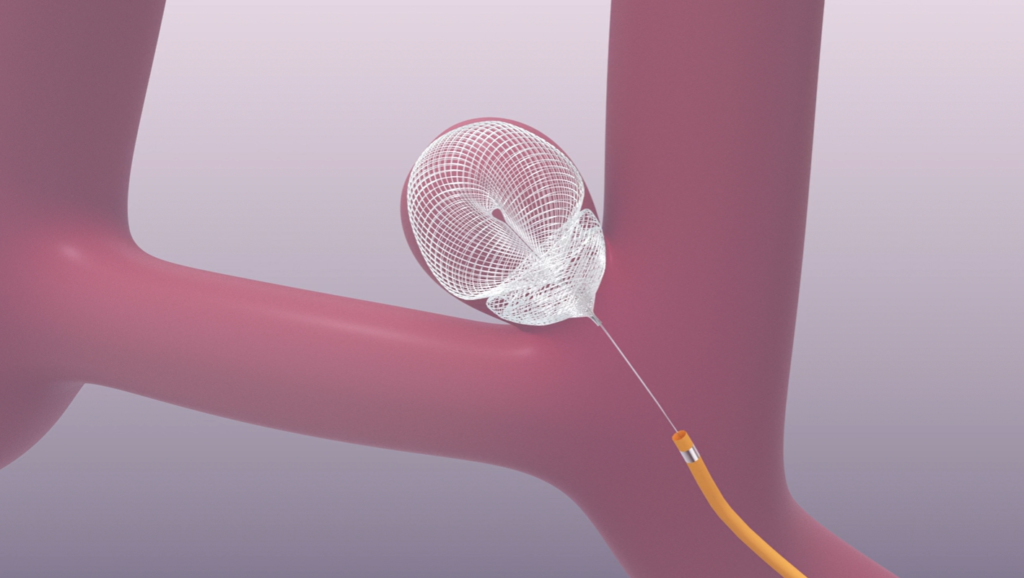
Non-clinical testing demonstrated that the SEALTM Embolization Device implant only is MR Conditional. It can be scanned safely under the following conditions:
Under the scan conditions defined above, the SEALTM Implant is expected to produce a maximum temperature rise of +1.4° C after 15 minutes of continuous scanning (i.e., per pulse sequence).
In non-clinical testing, the image artifact caused by the SEALTM Implant extends approximately 5 mm from the implant when imaged with a gradient echo pulse sequence and a 3-Tesla MRI system.
MR angiography may not be diagnostic due to the extent of the image artifact and alternate vascular imaging is recommended. The SEALTM Implant will create a signal loss depending on the pulse sequence parameters on a magnetic resonance angiography (MRA) exam, which may impact the diagnostic quality of the procedure. Therefore, an alternative imaging procedure may be needed, such as digital subtraction angiography (DSA).


For devices that will not be implanted: The Pusher Delivery System (non-implant) of the SEALTM Embolization System and the SEALTM Detachment Handle (non-implant) were not evaluated for magnetic resonance (MR) safety. It is classified as MR Unsafe.
Galaxy Therapeutics, INC.
799 Ames Avenue
Milpitas, CA 95035
Email: Contact@gtseal.com
